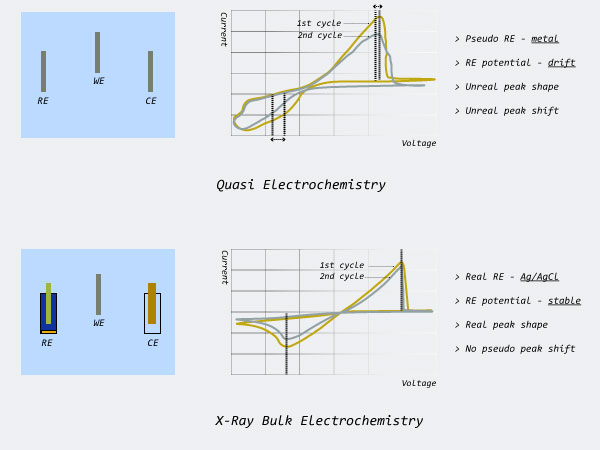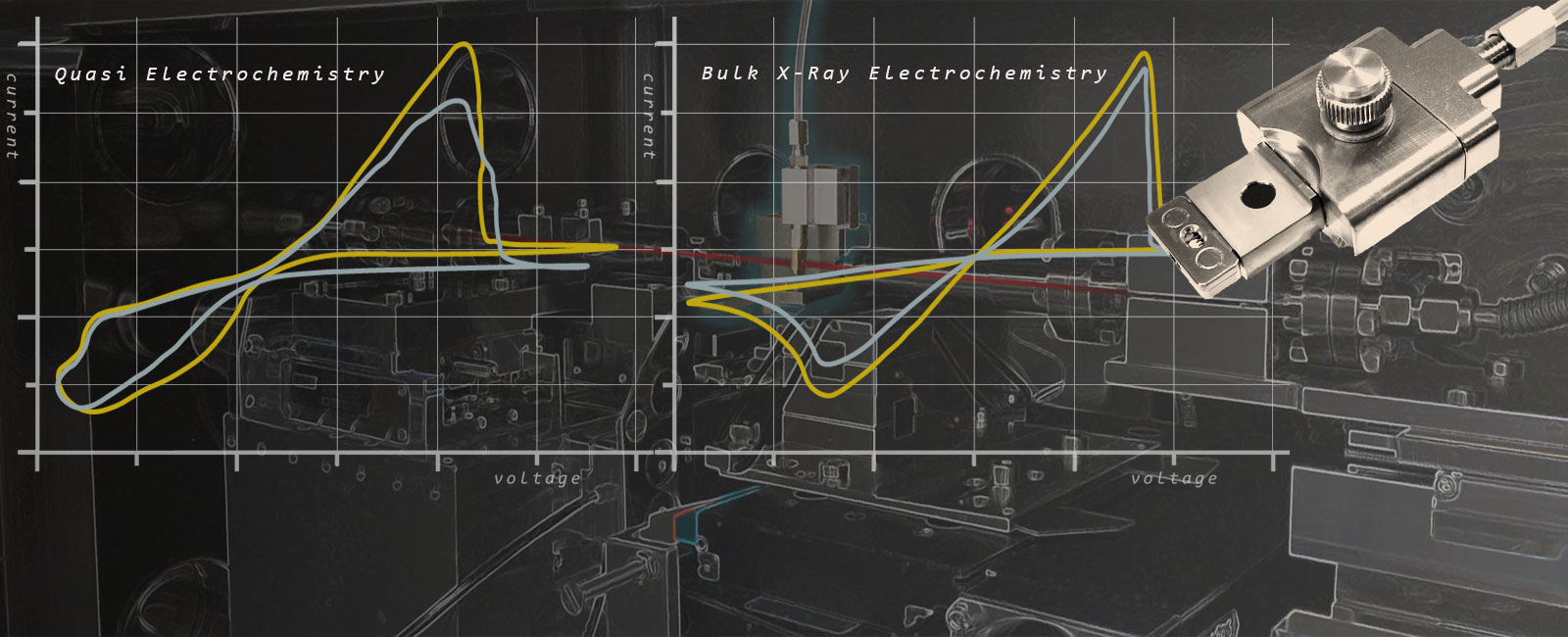

In-Situ X-Ray Bulk Liquid Electrochemistry
| 1470 Series X-Ray | |
| Total Electrodes | 6 |
| True Reference Electrode | Yes* – drift rate less than 0.1 mV/min |
| True Counter Electrode | Yes* |
| Electrolytes | Aqueous, Wide range of organics |
| Spacer Range | 100 nm to 50 μm* |
| Heating Compatibility | Yes |
| X-ray Microscope Compatibility | Custom integration |
Operando STXM demonstration of critical surface catalytic activity for hydrogen generation
William Chueh et al. from Stanford University, in collaboration with Hummingbird Scientific, Advanced Light Source (Lawrence Berkeley National Laboratory) and the University of Warwick used the operando STXM cell to demonstrate for the first time the surface site activity of transition metal (oxy) hydroxides with an electrochemical stimulus for developing a promising electrocatalyst for the oxygen evolution reaction (OER). This reaction is key in water splitting to generate hydrogen that can be used to store energy.
The data from the STXM electrochemical shows heterogeneity in the distribution of Co3+ species in the β-Co(OH)2 particles with an increase in the voltage. However, the electrochemical current is primarily restricted to the particle edge facets. The local concentration of higher Co oxidation state (Co3+) compared to the bulk of the particles suggests that layered oxides’ OER activity can be improved by improvising the surface morphology.
Figure: Cyclic voltammogram in 0.1M KOH of β-Co(OH)2 in the STXM electrochemical flow cell at a scan rate of 10 mV s-1 and a flow rate of 30 μL min-1. The voltage-dependent Co oxidation state phasemap of β-Co(OH)2 is also shown.
Image Copyright © 2021 Springer Nature Limited
Reference: Mefford et al., Nature, 2020 Full Paper
Edit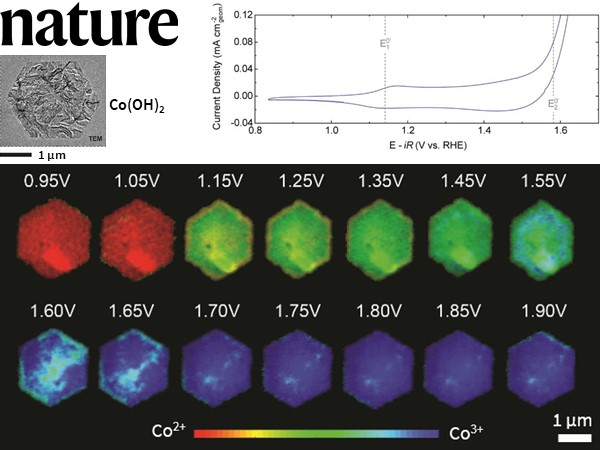
Computer vision aided modeling of reaction rates during operando battery cycling
Researchers at the Massachusetts Institute of Technology, Stanford University, and the Toyota Research Institute conducted in-situ scanning transmission X-ray microscopy (STXM) using their Hummingbird Scientific in-situ X-ray bulk liquid electrochemistry sample holder to study heterogeneous reaction kinetics in lithium iron phosphate (LFP) nanoparticles. The work combined a large data set of X-ray chemical mappings with computer vision to develop a data-driven model for heterogeneous reaction kinetics. This integrated and data-driven approach is critical for improving the efficiency of battery and electrocatalyst materials where spatially heterogeneous and unstable interfaces govern complex reaction kinetics.
Video: X-ray chemical maps of LiFePO4 nanoparticles during charging and discharging. Experimental STXM (left) and simulated results (right) show remarkable agreement. Scale bar 1 μm.
Video Copyright © 2023 Springer Nature Limited
Reference: Zhao et al. Nature 2023, 621, pp. 289-294. DOI: 10.1038/s41586-023-06393-x
Edit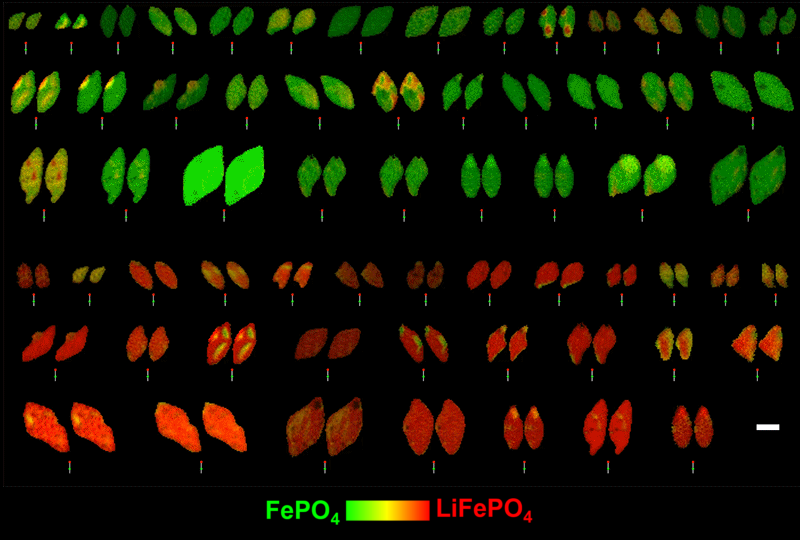
| Hongbo Zhao, Haitao Dean Deng, Alexander E. Cohen, Jongwoo Lim, Yiyang Li, Dimitrios Fraggedakis, Benben Jiang, Brian D. Storey, William C. Chueh, Richard D. Braatz, and Martin Z. Bazant. “Learning heterogeneous reaction kinetics from X-ray videos pixel by pixel.” Nature (2023) | Abstract | |
| Bonho Koo, Jinkyu Chung, Juwon Kim, Dimitrios Fraggedakis, Sungjae Seo, Chihyun Nam, Danwon Lee, Jeongwoo Han, Sugeun Jo, Hongbo Zhao, Neel Nadkarni, Jian Wang, Namdong Kim, Markus Weigand, Martin Z. Bazant, and Jongwoo Lim. “Dynamic surface phases controlling asymmetry of high-rate lithiation and delithiation in phase-separating electrodes.” Energy Environ. Sci. (2023) | Abstract | |
| J. Tyler Mefford, Andrew R. Akbashev, Minkyung Kang, Cameron L. Bentley, William E. Gent, Daan Hein Alsem, Norman Salmon, David A. Shapiro, Patrick R. Unwin, and William C. Chueh. “Correlative operando microscopy of oxygen evolution electrocatalysts.” Nature (2021) | Abstract | |
| David A. Shapiro, Sergey Babin, Richard S. Celestre, Weilun Chao, Raymond P. Conley,Peter Denes, Bjoern Enders, Pablo Enfedaque, Susan James, John M. Joseph, Harinarayan Krishnan, Stefano Marchesini, Krishna Muriki, Kasra Nowrouzi, Sharon R. Oh, Howard Padmore, Tony Warwick, Lee Yang, Valeriy V. Yashchuk, Young-Sang Yu, and Jiangtao Zhao. “An ultrahigh-resolution soft x-ray microscope for quantitative analysis of chemically heterogeneous nanomaterials.” Sci. Adv. (2020) | Abstract | |
| Mi Yoo, Young-Sang Yu, Hyunwoo Ha, Siwon Lee, Jin-Seok Choi, Sunyoung Oh, Eunji Kang, Hyuk Choi, Hyesung An, Kug-Seung Lee, Jeong Young Park, Richard Celestre, Matthew A. Marcus, Kasra Nowrouzi, Doug Taube, David A. Shapiro, WooChul Jung, Chunjoong Kim, and Hyun You Kim. “A tailored oxide interface creates dense Pt single-atom catalysts with high catalytic activity.” Energy & Environmental Science (2020) | Abstract | |
| J. Tyler Mefford, Khim Karki, Daan Hein Alsem, David Shapiro, Norman Salmon, and William C. Chueh. “Operando Scanning Transmission X-ray Microscopy of Co(OH)2 Oxygen Evolution Electrocatalysts.” Microscopy & Microanalysis (2019) | Abstract | |
| Xiaoyang Liu, Donald Vonk, Hua Jiang, Kim Kisslinger, Xiao Tong, Mingyuan Ge, Evgeny Nazaretski, Bruce Ravel, Kate Foster,Stanislas Petrash, and Yu-chen Karen Chen-Wiegart. “Environmentally Friendly Zr-Based Conversion Nanocoatings for Corrosion Inhibition of Metal Surfaces Evaluated by Multimodal X-ray Analysis.” ACS Appl. Nano Mater. (2019) | Abstract | |
| Khim Karki, Tyler Mefford, Daan Hein Alsem, Norman Salmon, and William C Chueh. “Replicating bulk electrochemistry in liquid cell microscopy.” Microscopy & Microanalysis (2018) | Abstract | |
| Jongwoo Lim, Yiyang Li, Daan Hein Alsem, Hongyun So, Sang Chul Lee, Peng Bai, Daniel A Cogswell, Xuzhao Liu, Norman Jin, Young-sang Yu, Norman J Salmon, David Shapiro, Martin Z Bazant, Tolek Tyliszczak, and William Chueh. “Using Scanning Transmission X-ray Microscopy to Reveal the Origin of Lithium Compositional Spatiodynamics in Battery Materials.” Microscopy & Microanalysis (2017) | Abstract | |
| J. Lim, Y. Li, D. H. Alsem, H. So, S. C. Lee, P. Bai, D.A. Cogswell, X. Liu, N. Jin, Y. Yu, N. J. Salmon, D. A. Shapiro, M. Z. Bazant, T.Tyliszczak, and W. C. Chueh. “Origin and Hysteresis of Lithium Compositional Spatiodynamics Within Battery Primary Particles.” Science (2016) | Abstract | |
| A. Kammers, D.H. Alsem, J. Lim, Y. Li, W. Chueh, and N. Salmon. “Accelerating Next Generation Battery Development Through the Application of Cross-Correlative In-Situ Microscopy.” Microscopy & Microanalysis (2015) | Abstract | |
| B. Stripe, V. Rose, M. Misek, S.W. Chee, D.H. Alsem, and N. Salmon. “Applications of In-Situ Synchrotron Radiation Techniques in Nanomaterials Research.” MRS Spring (2014) | Abstract |
Read More