Catalysis
| Gas-Heating | Bulk Liquid Electrochemistry | Heating-Biasing | TEM Tomography | ||
| Microscope/Technique |
TEM version available |  |
 |
 |
 |
| SEM version available |  |
 |
 |
 |
|
| X-Ray version available |  |
 |
 |
 |
|
| Stimuli |
Electrical |  |
 |
 |
 |
| Thermal |  |
 |
 |
 |
|
| Optical TEM version available |  |
 |
 |
 |
|
| Imaging | Higher resolution and diffraction |  |
 |
 |
 |
| EDS/EELS compatibility |  |
 |
 |
 |
|
| 3D reconstruction |  |
 |
 |
 |
|
| In-situ imaging |  |
 |
 |
 |
|
| Pre-and post-mortem analysis |  |
 |
 |
 |
|
| Environment |
Liquid |  |
 |
 |
 |
| Gas |  |
 |
 |
 |
|
| Vacuum |  |
 |
 |
 |
|
 Excellent
Excellent  Good
Good  N/A
N/A
Mapping of Catalytic Reactions in Real Time
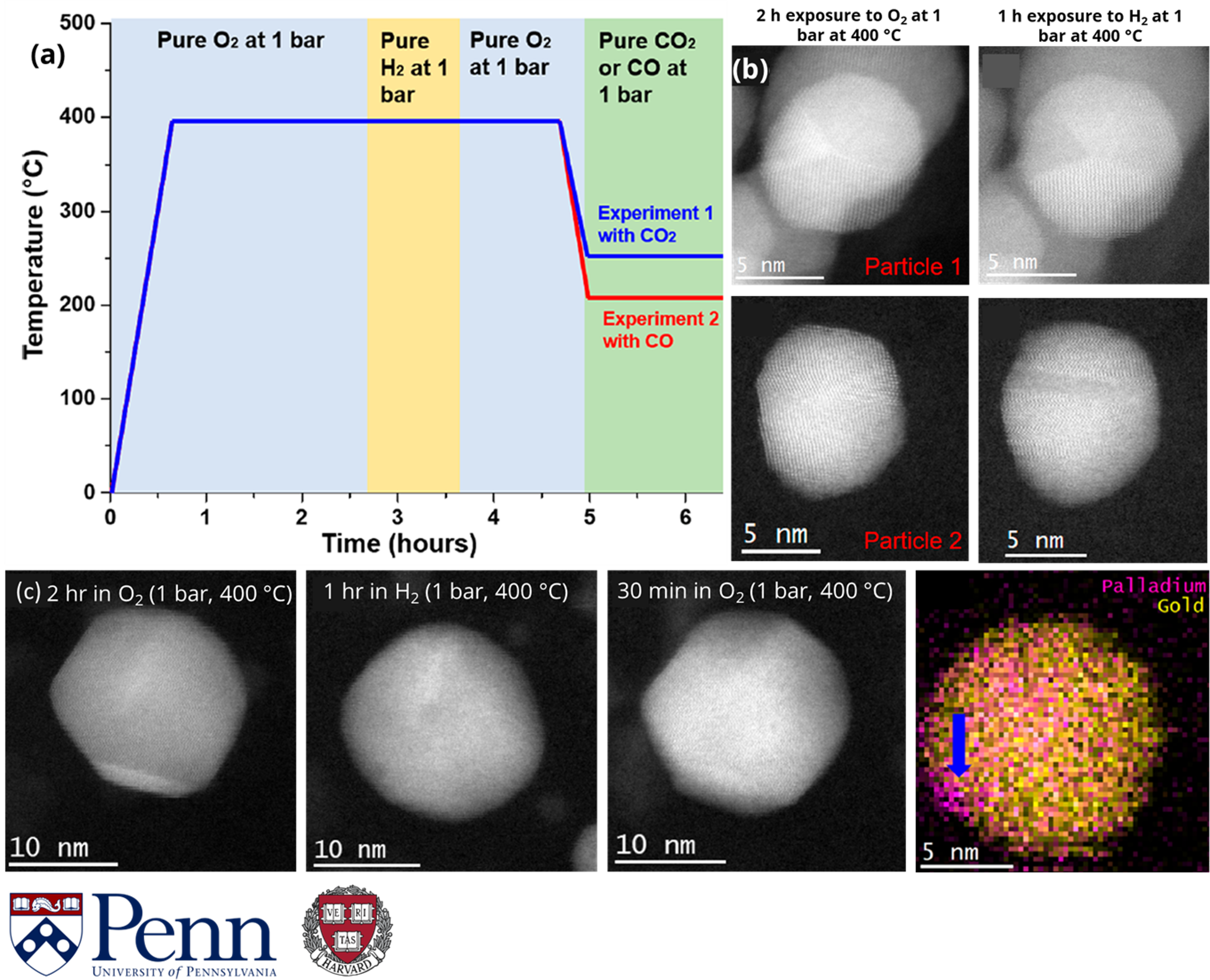
Researchers at Brookhaven National Laboratory have used Hummingbird Scientific in-situ gas cell TEM and X-Ray specimen holders to characterize heterogeneous catalytic reactions. By using correlated gas cell scanning transmission electron microscopy (STEM) and X-ray microscopy and spectroscopy they were able to quantitatively characterize catalytic activity of supported Pt catalysts. The capability of the Hummingbird Scientific gas cell to be transferred between the TEM and X-Ray microscope allowed a variety of probes to characterize a model catalytic reaction. This method can be broadly applied to study operando gas-reaction studies.
Reference: Y. Li, D. Zakharov, S. Zhao, R. Tappero, U. Jung, A. Elsen, Ph. Baumann, R.G. Nuzzo, E.A. Stach & A.I. Frenkel, “Complex structural dynamics of nanocatalysts revealed in Operando conditions by correlated imaging and spectroscopy probes”. Nature Communications (2015) Abstract
Edit

Operando reaction of Ferrihydrite nano-particles to Magnetite. Hydrogen gas at 1.1 bar was flown into the in-situ TEM gas cell during the experiment. When heated at 360°C the particles show changes from an amorphous to a crystalline structure as part of reducing reactions. The video shows this transformation. Imaging performance at temperature and pressure was explored and shown to almost match imaging performance of imaging in vacuum of similar samples on similar substrates. With the range of pressures and temperatures accessible with this experimental system, these types of experiments can be used to quantitatively explore kinetics over a large range of temperatures and pressures.
Hummingbird Scientific internal data in collaboration with Jaco Olivier, Matthew Coombes, Jan Neethling, Nelson Mandela Metropolitan University, South Africa
Edit| J.H. Nam, K.A. Mkhoyan, D.H. Alsem & P.J. Bruggeman, “Revealing the mechanisms of non-thermal plasma-enabled iron oxide reduction through nanoscale operando TEM”. Nat. Commun. (2025) | Abstract |
| Z. Manzoor, R. Karthik, M.A. Ferreira, D.S. Galvao, N.K. Mukhopadhyay, T.P. Yadav, P. Dadhwal, P.C. Saka, C.F. Woellner, S. Chowdhury, & C.S. Tiwary, “Radio frequency-induced catalysis using multi-component two-dimensional quasicrystals for effective sulfamethoxazole removal from water”. Appl. Catal. B (2025) | Abstract |
| E. Kang, J. Yun, H. Choi, M. Yoo, J.H. Lee, H. Park, J.-S. Choi, K.-S. Lee, D.A. Shapiro, A. Ditter, B.J. Kwon, C. Kim, Y.-S. Yu & H.Y. Kim, “Pt Nanoparticle Disintegration at Oxide Interfaces Enhances CO Oxidation Catalysis”. Small (2025) | Abstract |
| G.G. Hollyer, D.N. Zakharov, D.H. Alsem, C. Parkin & E.A. Stach, “Nanometer-scale resolution in an ultra-high pressure environmental TEM holder”. MRS Commun. (2025) | Abstract |
| A.C. Meng, P. Cheng, P.R. Kidambi, N. Singh, Y. Song, & E.A. Stach, “Characterization of iron-based alloy catalysts for atmospheric pressure chemical vapor deposition of carbon nanofibers via in situ transmission electron microscopy”. J. Vac. Sci. Technol. B (2025) | Abstract |
| X. Lu, S. Hwang & K. He, “In Situ TEM for Structural and Chemical Evolutions of Bimetallic Pt–Ni Nanoparticles at Elevated Temperatures: Implications for Heterogeneous Catalysis”. ACS Appl. Nano Mater. (2024) | Abstract |
| A.C. Foucher & E.A. Stach, “Identifying dynamic restructuring effects in nanocatalysts by combining in situ scanning transmission electron microscopy and in situ X-ray absorption spectroscopy: A review”. Catal. Today (2024) | Abstract |
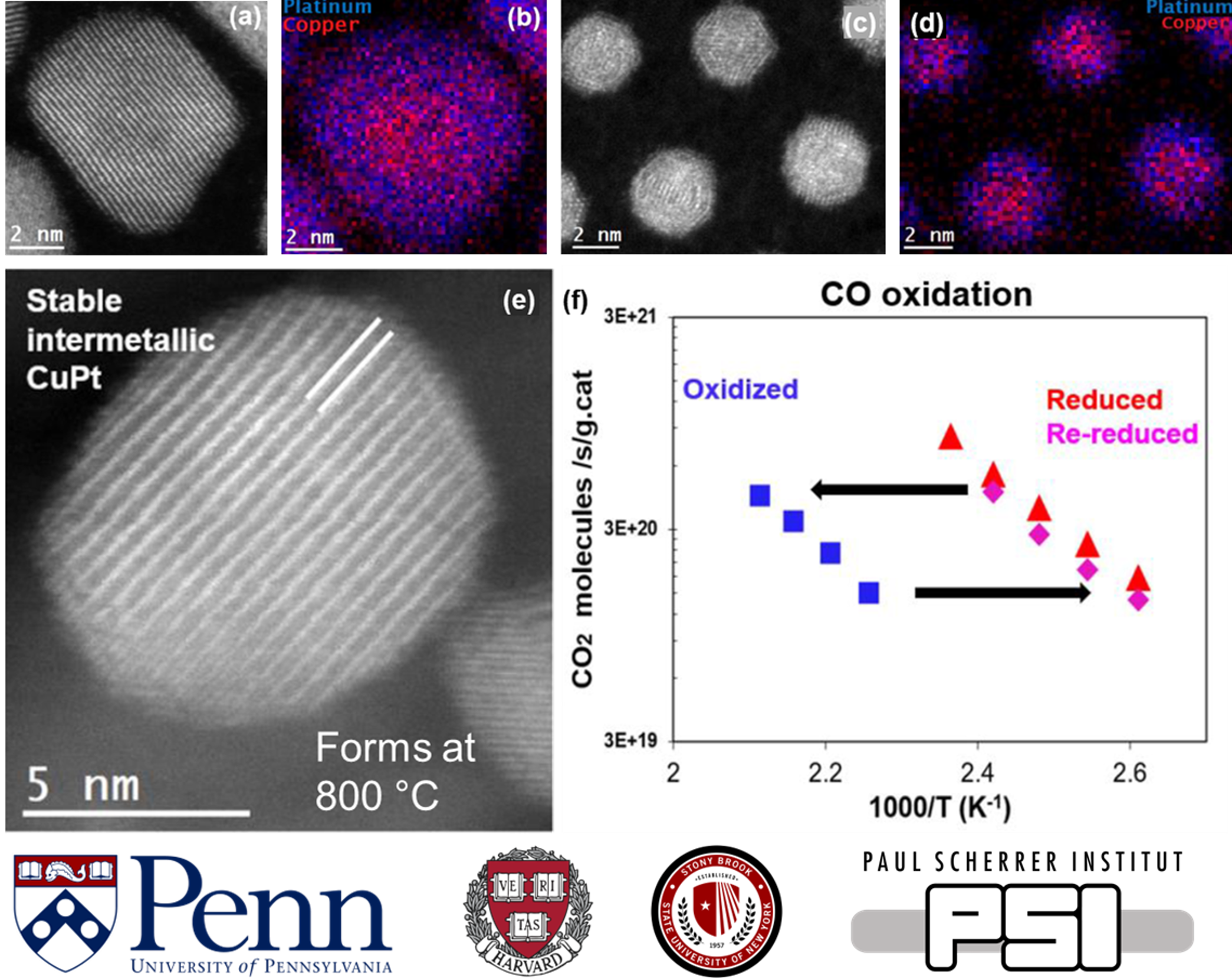
| A.C. Foucher, S. Yang, D.J. Rosen, R. Huang, J.B. Pyo, O. Kwon, C.J. Owen, D.F. Sanchez, I.I. Sadykov, D. Grolimund, B. Kozinsky, A.I. Frenkel, R.J. Gorte, C.B. Murray & E.A. Stach, “Synthesis and Characterization of Stable Cu–Pt Nanoparticles under Reductive and Oxidative Conditions”. J. Am. Chem. Soc. (2023) | Abstract |
| J.P. Horwath, C. Lehman-Chong, A. Vojvodic & E.A. Stach, “Surface Rearrangement and Sublimation Kinetics of Supported Gold Nanoparticle Catalysts”. ACS Nano (2023) | Abstract |
| Z.-Y. Wu, P. Zhu, D.A. Cullen, Y. Hu, Q.-Q. Yan, S.-C. Shen, F.-Y. Chen, H. Yu, M. Shakouri, J.D. Arregui-Mena, A. Ziabari, A.R. Paterson, H.-W. Liang & H. Wang, “A general synthesis of single atom catalysts with controllable atomic and mesoporous structures”. Nat. Synth. (2022) | Abstract |
| J.D. Lee, J.B. Miller, A.V. Shneidman, L. Sun, J.F. Weaver, J. Aizenberg, J. Biener, J.A. Boscoboinik, A.C. Foucher, A.I. Frenkel, J.E.S. van der Hoeven, B. Kozinsky, N. Marcella, M.M. Montemore, H.T. Ngan, C.R. O’Connor, C.J. Owen, D.J. Stacchiola, E.A. Stach, R.J. Madix, P. Sautet & C.M. Friend, “Dilute Alloys Based on Au, Ag, or Cu for Efficient Catalysis: From Synthesis to Active Sites”. Chem. Rev. (2022) | Abstract |
| A.C. Foucher, J.D. Lee, Z. Qi, G. Li, G. Ouyang, J. Cui, J.A. Boscoboinik, C.M. Friend, J. Biener & E.A. Stach, “Boosting the H2 –D2 Exchange Activity of Dilute Nanoporous Ti–Cu Catalysts through Oxidation–Reduction Cycle–Induced Restructuring”. Adv. Eng. Mater. (2022) | Abstract |
| J.D. Lee, Z. Qi, A.C. Foucher, H.T. Ngan, K. Dennis, J. Cui, I.I. Sadykov, E.J. Crumlin, P. Sautet, E.A. Stach, C.M. Friend, R.J. Madix & J. Biener, “Facilitating Hydrogen Dissociation over Dilute Nanoporous Ti–Cu Catalysts”. J. Am. Chem. Soc. (2022) | Abstract |
| A.C. Meng, K.-B. Low, A.C. Foucher, Y. Li, I. Petrovic & E.A. Stach, “Anomalous metal vaporization from Pt/Pd/Al2O3 under redox conditions”. Nanoscale (2021) | Abstract |
| M. Luneau, E. Guan, W. Chen, A.C. Foucher, N. Marcella, T. Shirman, D.M.A. Verbart, J. Aizenberg, M. Aizenberg, E.A. Stach, R.J. Madix, A.I. Frenkel & C.M. Friend, “Enhancing catalytic performance of dilute metal alloy nanomaterials”. Commun. Chem. (2020) | Abstract |
| B. Song, Y. Yang, M. Rabbani, T.T. Yang, K. He, X. Hu, Y. Yuan, P. Ghildiyal, V.P. Dravid, M.R. Zachariah, W.A. Saidi, Y. Liu & R. Shahbazian-Yassar, “In Situ Oxidation Studies of High-Entropy Alloy Nanoparticles”. ACS Nano (2020) | Abstract |
| M. Yoo, Y.-S. Yu, H. Ha, S. Lee, J.-S. Choi, S. Oh, E. Kang, H. Choi, H. An, K.-S. Lee, J.Y. Park, R. Celestre, M.A. Marcus, K. Nowrouzi, D. Taube, D.A. Shapiro, W. Jung, C. Kim & H.Y. Kim, “A tailored oxide interface creates dense Pt single-atom catalysts with high catalytic activity”. Energy Environ. Sci. (2020) | Abstract |
| Chen Houa, Jiuhui Hanb, Pan Liua, Chuchu Yangb, Gang Huangb, Takeshi Fujitab, Akihiko Hiratab, and Mingwei Chen. “Operando observations of RuO2 catalyzed Li2O2 formation and decomposition in a Li-O2 micro-battery,” Nano Energy (2018) | Abstract |
| S.D. House, J.J. Vajo, C. Ren, N.J. Zaluzec, A.A. Rockett & I.M. Robertson, “Impact of initial catalyst form on the 3D structure and performance of ball-milled Ni-catalyzed MgH2 for hydrogen storage”. Int. J. Hydrogen Energy (2017) | Abstract |
| Yimin A. Wu, Liang Li, Zheng Li, Alper Kinaci, Maria K. Y. Chan, Yugang Sun, Jeffrey R. Guest, Ian McNulty, Tijana Rajh, and Yuzi Liu. “Visualizing Redox Dynamics of a Single Ag/AgCl Heterogeneous Nanocatalyst at Atomic Resolution,” ACS Nano (2016) | Abstract |
| S.D. House, J.J. Vajo, C. Ren, A.A. Rockett & I.M. Robertson, “Effect of ball-milling duration and dehydrogenation on the morphology, microstructure and catalyst dispersion in Ni-catalyzed MgH2 hydrogen storage materials”. Acta Mater. (2015) | Abstract |
| Y. Li, D. Zakharov, S. Zhao, R. Tappero, U. Jung, A. Elsen, Ph. Baumann, R.G. Nuzzo, E.A. Stach & A.I. Frenkel, “Complex structural dynamics of nanocatalysts revealed in Operando conditions by correlated imaging and spectroscopy probes”. Nature Communications (2015) | Abstract |
| H.L. Xin, K. Niu, D.H. Alsem, and H. Zheng. “In-Situ TEM Study of Catalytic Nanoparticle Reactions in Atmospheric Pressure Gas Environment,” Microscopy and Microanalysis (2013) | Abstract |
| J.C. Yang, M.W. Small, R.V. Grieshaber & R.G. Nuzzo, “Recent developments and applications of electron microscopy to heterogeneous catalysis”. Chem. Soc. Rev. (2012) | Abstract |
| K.L. Jungjohann, J.E. Evans, J.A. Aguiar, I. Arslan & N.D. Browning, “Atomic-Scale Imaging and Spectroscopy for In Situ Liquid Scanning Transmission Electron Microscopy”. Microsc. Microanal. (2012) | Abstract |
| S.M. Kim, C.L. Pint, P.B. Amama, R.H. Hauge, B. Maruyama, E.A. Stach. “Catalyst and catalyst support morphology evolution in single-walled carbon nanotube supergrowth: Growth deceleration and termination,” Journal of Material Resolution (2010) | Abstract |
Read More