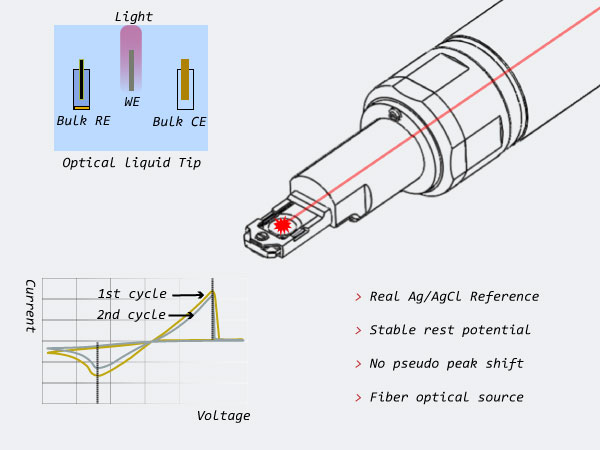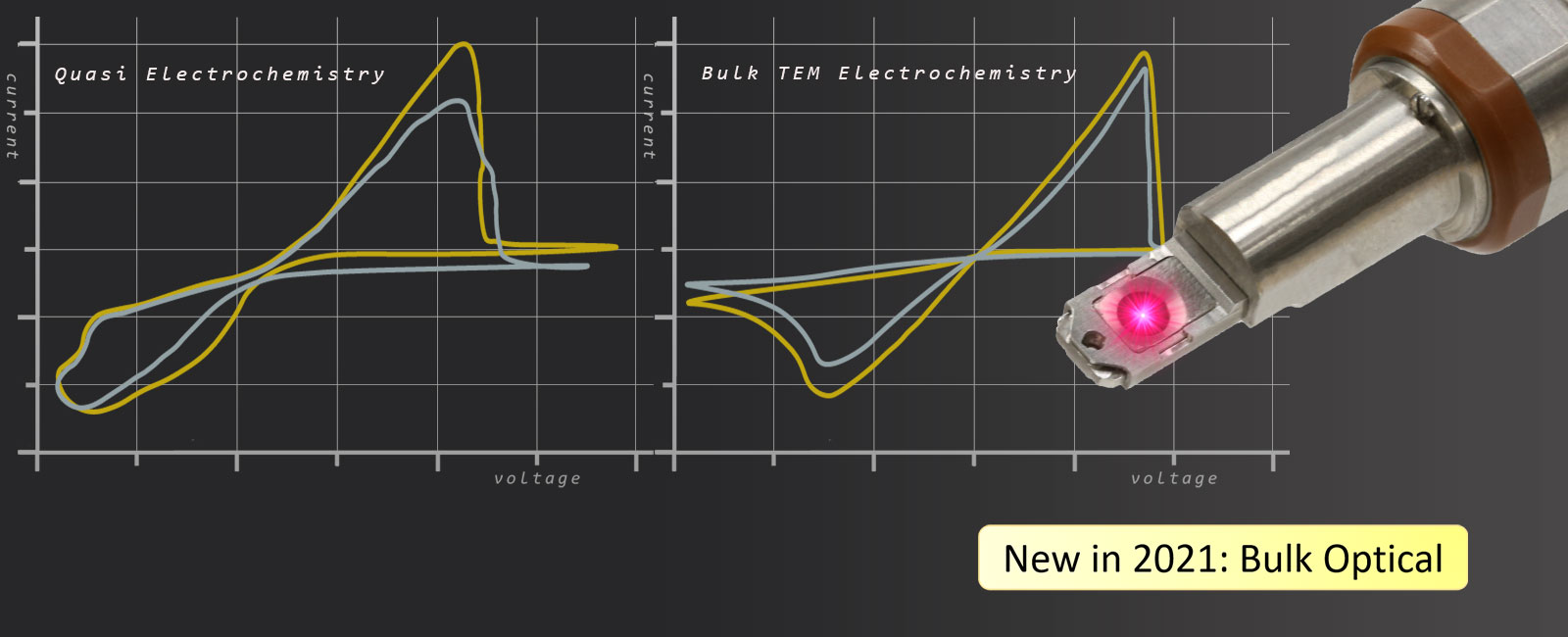

Optical Bulk Liquid Electrochemistry

| 1405 series | |
| Number of Inlets | 1 or 2 depending on model, single outlet |
| Total Electrodes | 6 |
| Fiber Bandwidth | 100 to 2000 nm* |
| True Reference Electrode | Yes* |
| True Counter Electrode | Yes* |
| Electrolytes | Aqueous, Wide range of organics |
| Spacer Range | 100 nm to 2 μm* |
| EELS/EDS Compatible | Yes |
| Heating Capability | Yes |
| TEM Compatibility | TFS/FEI, JEOL, Hitachi |
2D materials for photoelectrochemical water-splitting
Photoelectrochemistry provides a promising, environmentally friendly route to hydrogen production; however, the atomic-scale mechanisms of the photocatalysts that facilitate the water-splitting reaction are currently poorly understood. Researchers at the University of Pennsylvania have studied the photocatalytic activity of several 2D nanomaterials such as gold nanoprisms and MoS2 flakes. The research focused on the correlation between I-V characteristics with water splitting and simultaneous structural changes at the catalytically active sites. The I-V characteristic curve with the illuminated catalyst shows additional activity in the oxidation regime when operating in in-situ illuminated conditions. In contrast, such activities are not observed in the absence of the catalytic particles. TEM images show the formation of gas bubbles induced at the working electrode with the illumination of the catalysts corresponding to water splitting.
Image Right – Top: Schematic shows the optical setup in liquid cell and redox IV characteristics of active catalytic material with and without light. Bottom: In-situ TEM images show corresponding hydrogen evolution and bubble formation at the electrode-electrolyte interface during the hydrogen evolution reaction (HER). From left to right, the evolution of bubble formation is captured as a function of time and applied potential.
HBS internal data obtained in collaboration with Pawan Kumar, Deep Jariwala and Eric Stach at the University of Pennsylvania.
Edit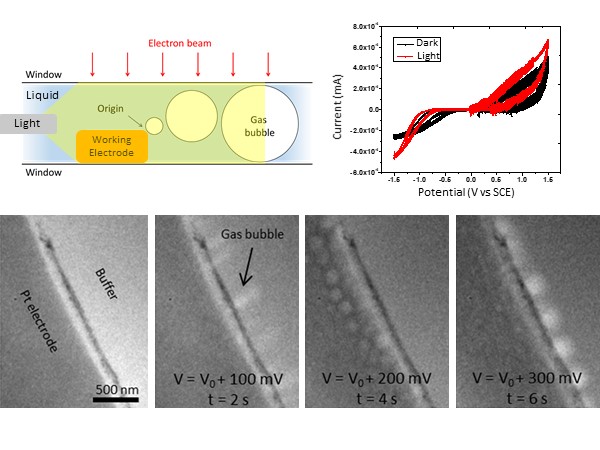
In-situ photocatalytic activity of molybdenum disulfide and hydrogen evolution reaction (HER)
The video on the left acquired as part of a collaborative project with the research groups of Deep Jariwala, and Eric Stach at the University of Pennsylvania shows a powered electrode in water. The electrode is part of a water-splitting experiment resulting in gas bubble formation, which is characteristic of gas evolution during the hydrogen evolution reaction (HER) at a working electrode (WE).
The evolution of hydrogen gas in this experiment is enhanced by the photocatalytic action of a two-dimensional (2D) molybdenum disulfide (MoS2) at the active electrode site illuminated by a white light source in Hummingbird Scientific’s in-situ Optical Bulk Liquid Electrochemistry TEM holder. The bulk reference electrode (RE) in the holder allows for an accurate representation of the HER, matching electrochemical behavior seen in the bulk, beaker-scale geometry of a similar reaction.
Edit| Khim Karki, Pawan Kumar, Antoine Verret, Noah Glachman, Daan Hein Alsem, Deep Jariwala, Norman Salmon, and Eric Stach. “In situ/operando Study of Photoelectrochemistry Using Optical Liquid Cell Microscopy,” Microscopy and Microanalysis (2020) | Abstract |
| Noah Glachman, Noah Geller, Alexander Shea, V. Antoine Verret, Khim Karki, Julio Rodriquez-Manzo, Norman J. Salmon, Daan Hein Alsem, Deep Jariwala and Eric Stach. “Development of a Method to Characterize Active Sites in Photocatalysis using operando Transmission Electron Microscopy,” Microscopy & Microanalysis (2019) | Abstract |
| Noah Glachman, Noah Geller, Alexander Shea, Khim Karki, Daan Hein Alsem, Deep Manoj Jariwala, Eric A. Stach. “Light-driven Liquid Cell Transmission Electron Microscopy to Study Photocatalytic Materials,” Materials Research Society Meeting (2019) | Abstract |
| Julio A. Rodriguez Manzo, Norman J. Salmon, and Daan Hein Alsem. “In Situ Observation of Water Splitting,” Microscopy & Microanalysis (2017) | Abstract |
| Khim Karki, Julio Alejandro Rodriguez Manzo, Daan Hein Alsem, and Norman Salmon. “Transmission Electron Microscopy Study of Bubble Formation at Metallic Electrodes in Liquid Environment,” Materials Research Society Meeting (2017) | Abstract |
Read More