Researchers at Harvard University, University of Pennsylvania, Stony Brook University, and Brookhaven National Laboratory studied dilute alloys for their activity for catalytic hydrogenation of 1-hexyne to 1-hexene. As part of this research, they showed, using a Hummingbird Scientific in-situ TEM gas-cell heating sample holder, that Palladium-Gold nanoparticles embedded in porous SiO2 remain structurally unchanged up to reaction-relevant high temperatures. Because these nanoparticles do not change structure during catalytic cycling, it shows these dilute metal alloy nanoparticles would be excellent catalysts. They published their work in Communications Chemistry.
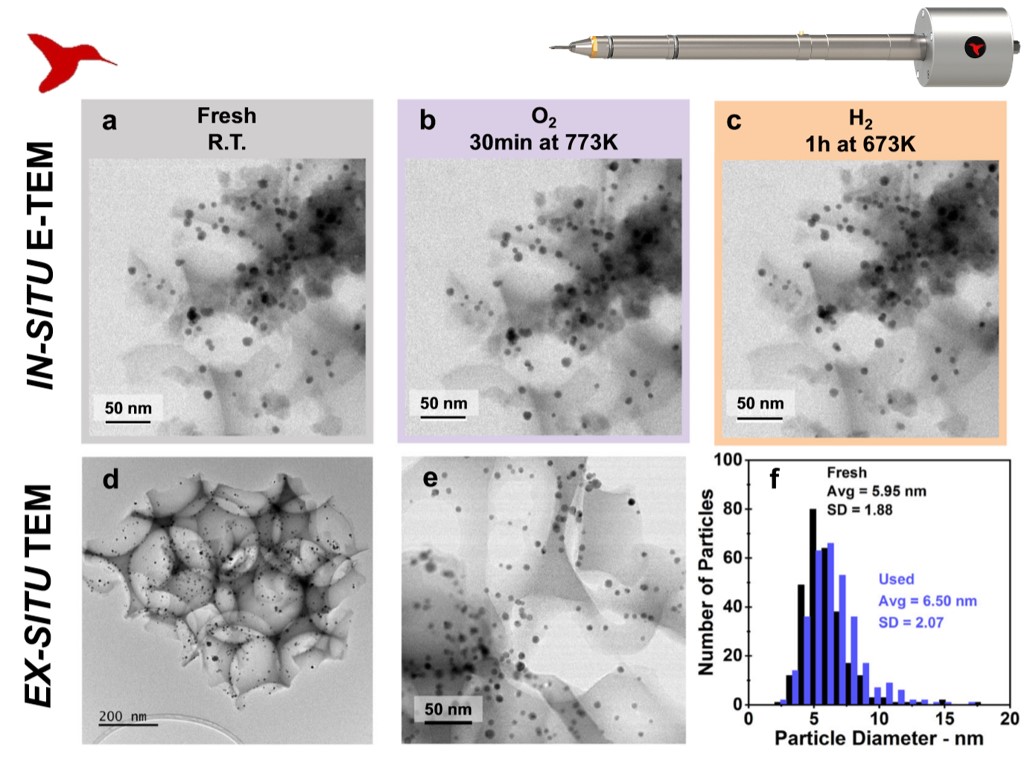
In-situ TEM shows that the silica structure and the shape of as-prepared Pd0.04Au0.96 RCT-SiO2 catalyst (a) do not alter when exposed to oxygen at 500C (b) and to hydrogen at 400C (c). Ex-situ TEM (d) shows that the silica structure is intact and the particles do not sinter after more than 10 catalytic cycles (e), (f) . From Communications Chemistry under the Creative Commons license.
Reference: M. Luneau, E. Guan, W. Chen, A. C. Foucher, N. Marcella, T. Shirman, D. M. A. Verbart, J. Aizenberg, M. Aizenberg, E. A. Stach, R. J. Madix, A. I. Frenkel, C. M. Friend, Enhancing catalytic performance of dilute metal alloy nanomaterials, Communications Chemistry, 3, 46 (2020) Full paper
View All News