What if you could visualize corrosion as it initiates and grows—inside your SEM?
Morgan Barbey-Binggeli and Vasiliki Tileli from EPFL published recent work using Hummingbird Scientific In-Situ SEM bulk liquid-electrochemistry sample holder to directly visualize localized anodic corrosion of aluminum in real time at the solid–liquid interface. By combining electrochemical control with operando SEM imaging in liquid, the study reveals how pitting initiates and evolves dynamically, including associated gas generation and changes in pit morphology.
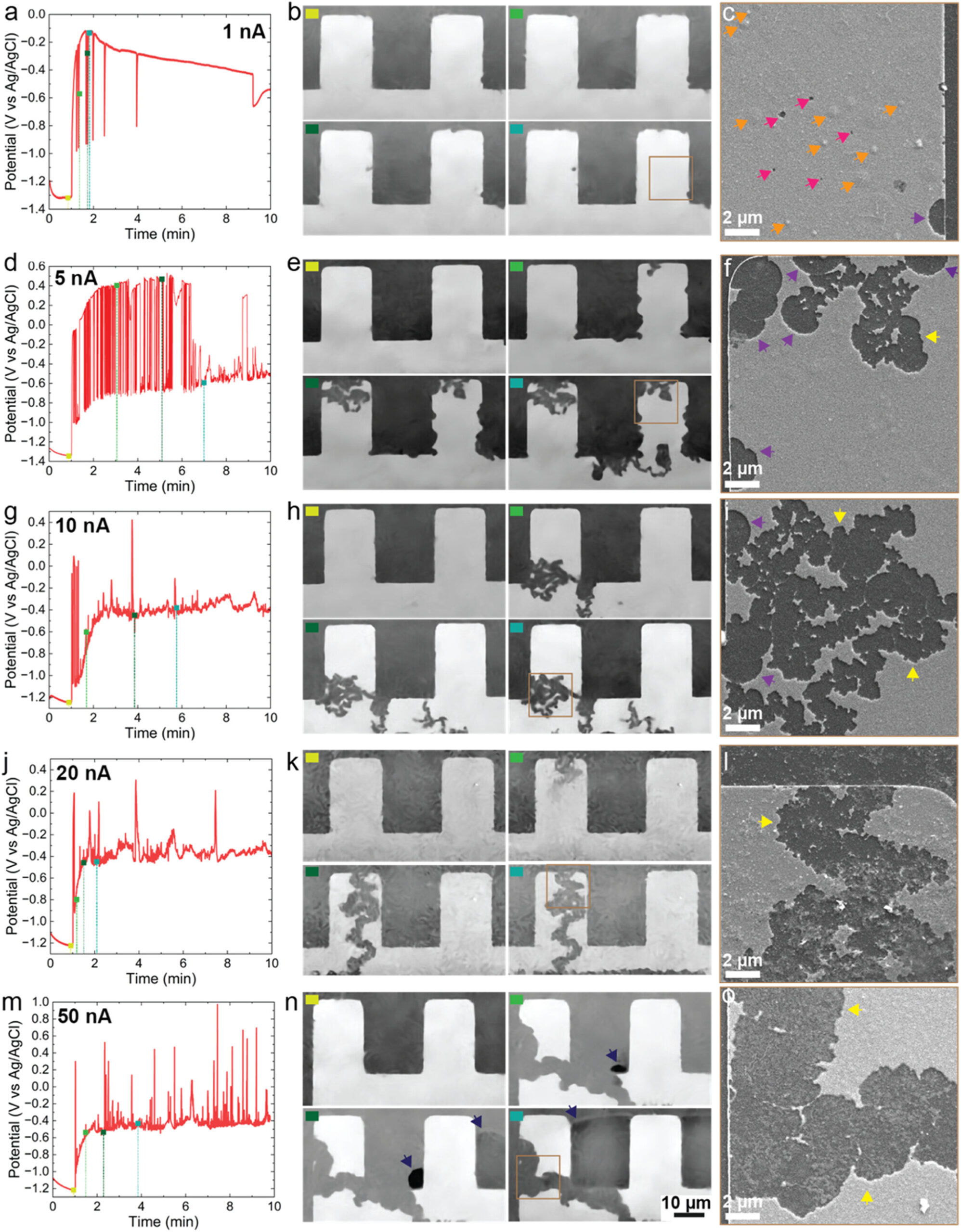
Time-resolved chronopotentiometry (CP) experiments were carried out in the SEM at 1, 5, 10, 20, and 50 nA. For each current condition, the electrochemical response is shown on the left, followed by in situ SEM images captured at selected time points in the middle, and post-mortem SEM images of the same region on the right. The colored marker in the top-left corner of the in situ images (b, e, h, k, n) matches the corresponding time points on the electrochemical curves (a, d, g, j, m). Areas outlined in brown in (b, e, h, k, n) indicate the locations later examined in the post-mortem images (c, f, i, l, o). In the post-mortem SEM images, magenta arrows mark pits, orange arrows mark intact blisters, purple arrows indicate circular corrosion features, and yellow arrows highlight fractal-like corrosion patterns. In (n), dark blue arrows show the formation and growth of a gas bubble.
In the experiments, the team used liquid-phase liquid-phase SEM and TEM workflows to monitor thin-film aluminum in saline electrolyte while applying galvanostatic (constant-current) conditions to drive corrosion in a controlled manner. As the applied current increased, corrosion progressed from early surface blisters and nanoscale pit nucleation to fully developed pit networks, including transitions from rounded pits to complex fractal-like dissolution fronts. Importantly, the operando imaging also enabled direct observation of gas bubble formation during advanced pitting, and follow-up analysis confirmed the evolved gas as molecular hydrogen (H₂).
This study highlights what becomes possible with the Hummingbird Scientific In-Situ SEM bulk liquid-electrochemistry holder: stable bulk electrochemistry inside the SEM paired with direct imaging in liquid, enabling researchers to connect electrochemical conditions to real-time interfacial processes that are otherwise difficult to capture. Beyond corrosion, this platform supports a wide range of solid–liquid electrochemical investigations, offering a powerful new route to uncover mechanisms and accelerate discovery across materials and electrochemical systems.
Reference: Morgan Barbey-Binggeli & Vasiliki Tileli, J. Am. Chem. Soc., 147(38), 34920–34932 (2025). DOI: 10.1021/jacs.5c11352
Full paper Copyright © 2025 The Authors. Published by American Chemical Society.
View All News