Lithium-oxygen (Li-O2) batteries have been actively pursued as a replacement for traditional lithium-ion systems because of their exceptionally higher energy densities suitable for long-range EVs and other applications. However, the byproduct of Li-O2 cells, Li2O2, cannot be easily decomposed during the charging cycle. The large overpotential during the charging provides a major challenge for the practical applications. Now, the researchers from Tohoku University, Shanghai Jiao Tong University and Johns Hopkins University have demonstrate that decomposition of Li2O2 can be improved by adding redox mediators as charge-transfer agents. Their findings are published in the recent issue of Advanced Materials.
Copyright © 2017 Wiley-VCH
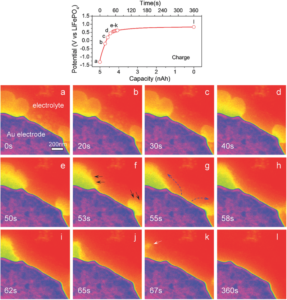
Using the Hummingbird TEM Liquid Electrochemistry holder, the researchers were able assemble a liquid-cell Li-O2 microbattery with LiFePO4 as a counter electrode (Li source) and gold electrode as a working electrode. Both electrodes were flooded with a continuous flow of an electrolyte solution consisting of 1M lithium percholate (LiClO4) dissolved in dimethyl sulfoxide (DMSO) with 50 x 10-3 M TTF redox mediator.
During the discharge cycle, there is a nanoscale formation of Li2O2 (bright yellow contrast in the image above) in the electrolyte. Upon the charge cycle, the Li2O2 phase decompose (arrows in the image above showing the reaction front), assisted by the redox mediator TFF at the liquid/solid interfaces of electrolyte/electrode and electrolyte/Li2O2. The operando liquid TEM cell provides direct experimental insights into the formation and decomposition of Li2O2 in the DMSO-based electrolyte and TTF redox mediators. The fundamental understanding of the Li-O2 electrochemistry presented in this work may enable the rapid development of Li-O2 batteries for practical applications.
Reference: Chuchu Yang, Jiuhui Han, Pan Liu, Chen Hou, Gang Huang, Takeshi Fujita, Akihiko Hirata, and Mingwei Chen. “Direct Observations of the Formation and Redox-Mediator-Assisted Decomposition of Li2O2 in a Liquid-Cell Li–O2 Microbattery by Scanning Transmission Electron Microscopy,” Advanced Materials (2017). DOI:10.1002/adma.201702752
View All News

