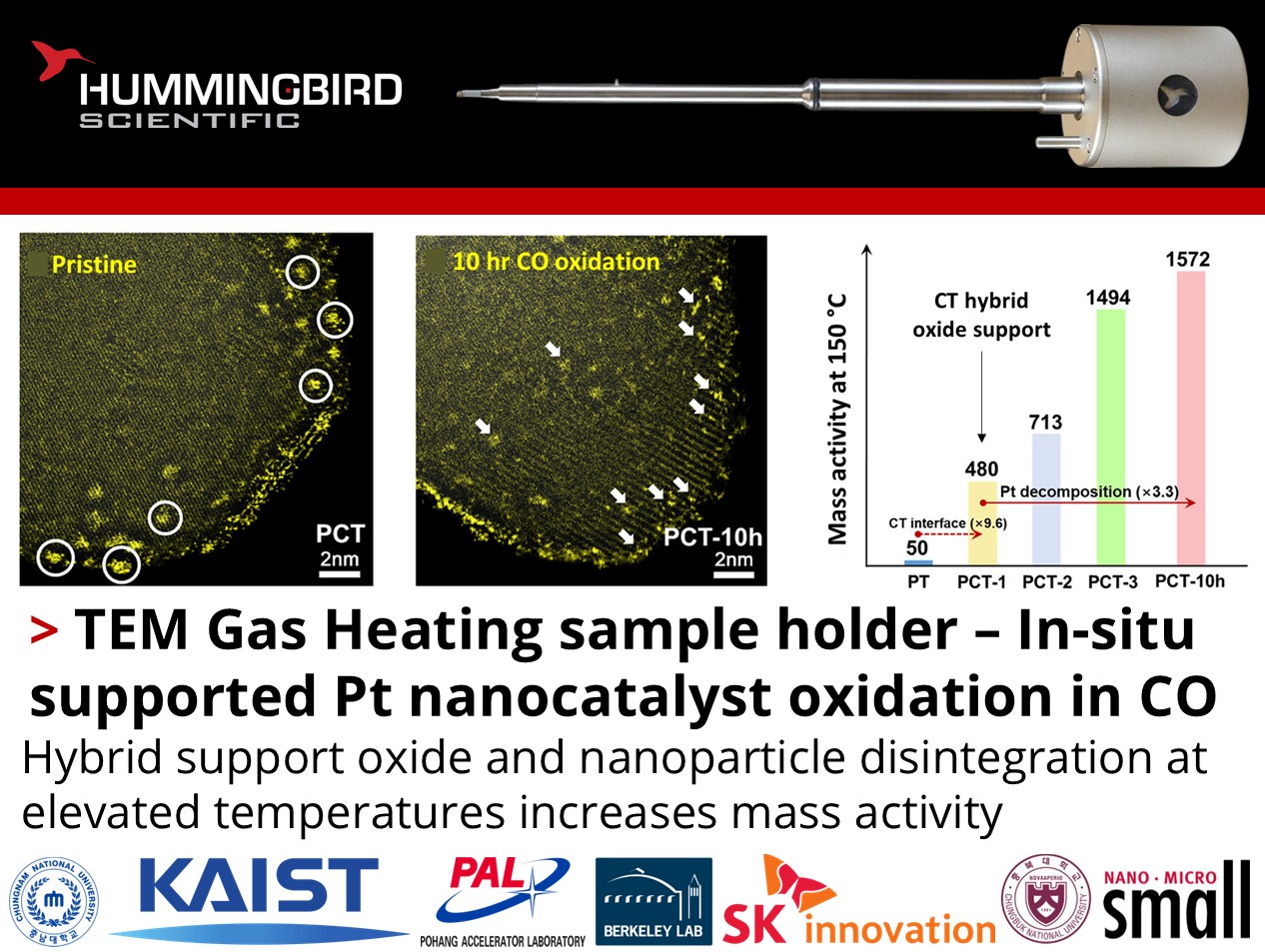
How can the influence of nanoparticle support structures and disintegration on catalytic activity be directly observed?
Eunji Kang and Hyun You Kim from Chungnam National University, along with their colleagues from Korea Advanced Institute of Science and Technology, Pohang Accelerator Laboratory, Berkeley Lab, SK Innovation, and Chungbuk National University published recent work using the Hummingbird Scientific TEM gas heating sample holder and X-ray gas heating sample holder to characterize the Pt nanoparticle disintegration dynamics on CeOx-TiO2 (CT) hybrid support oxides during carbon monoxide (CO) oxidation. In-situ scanning transmission electron microscopy (STEM) was combined with density functional theory (DFT) calculations, in-situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS), X-ray absorption spectroscopy (XAS) and scanning transmission X-ray microscopy (STXM) to reveal the mechanism behind dynamic disintegration of Pt nanoparticles into single atoms (SAs) or sub-nanometer clusters.
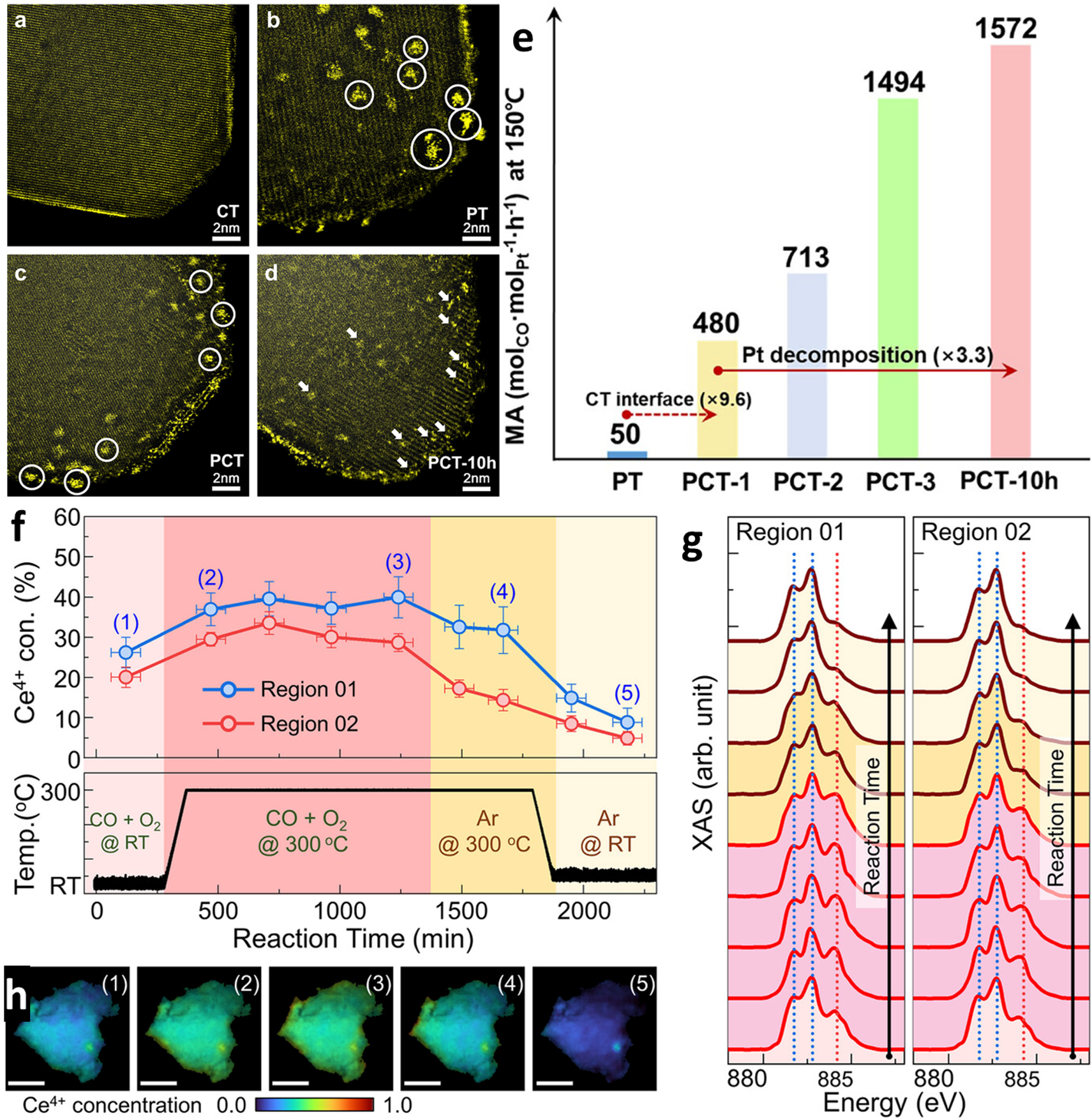
HAADF-STEM images of the hybrid supporting oxide a) and the resultant Pt-containing catalysts b–d). a) CT hybrid supporting oxide, b) PT, c) PCT, and d) PCT after 10 h of CO oxidation reaction. White circles in (b,c) present Pt NPs. White arrows in (d) highlight Pt-SAs and clusters. e) Enhanced specific mass activity of PCT during repeated and prolonged CO oxidation reaction. The PCT-n presents the number of repeating historical cycles of CO oxidation to which the PCT catalyst is exposed. The MA of Pt was increased by 3.3 times from PCT-1 to PCT-10 h. The MA of PT catalyst without Ce addition was increased by 9.6 times upon adding 2 wt.% of Ce (from PT to PCT-1). f) In-situ tracking of Ce4+ concentration and temperature for the in situ XAS and STXM CO oxidation (1 vol.% CO + 4 vol.% O2 + 95 vol.% Ar) experiment. g) Averaged XAS spectra for two the experimental regions and h) STXM Ce oxidation state maps under the conditions marked (1)-(5) in (f). Copyright © 2025 The Author(s). Small published by Wiley-VCH GmbH
In-situ STEM characterization revealed morphological changes to supported Pt nanoparticles while in-situ spectroscopic analyses confirmed oxygen-driven disintegrative transformation correlated to a threefold increase in mass-specific activity over 10 hours of CO oxidation at 150 °C. Compared to single oxide TiO2-supported Pt nanoparticles, the hybrid oxide support also facilitated a nearly tenfold increase in mass-specific activity over one cycle. DFT indicated weakened Pt-Pt cohesion in the presence of CO, while important reaction intermediates were stabilized at the CeOx interface. These findings demonstrate an opportunity to leverage interfacial oxygen dynamics and metal-support interactions to optimize catalytic performance with favorable nanoparticle disintegration pathways.
Reference: Eunji Kang, Jieun Yun, Hyuk Choi, Mi Yoo, Ju Hyeok Lee, Hongjin Park, Jin-Seok Choi, Kug-Seung Lee, David A. Shapiro, Alex Ditter, Bob Jin Kwon, Chunjoong Kim, Young-Sang Yu, Hyun You Kim, Small 21 (52) (2025) DOI:10.1002/smll.202506990
Full paper Copyright © 2025 The Author(s). Small published by Wiley-VCH GmbH
View All News